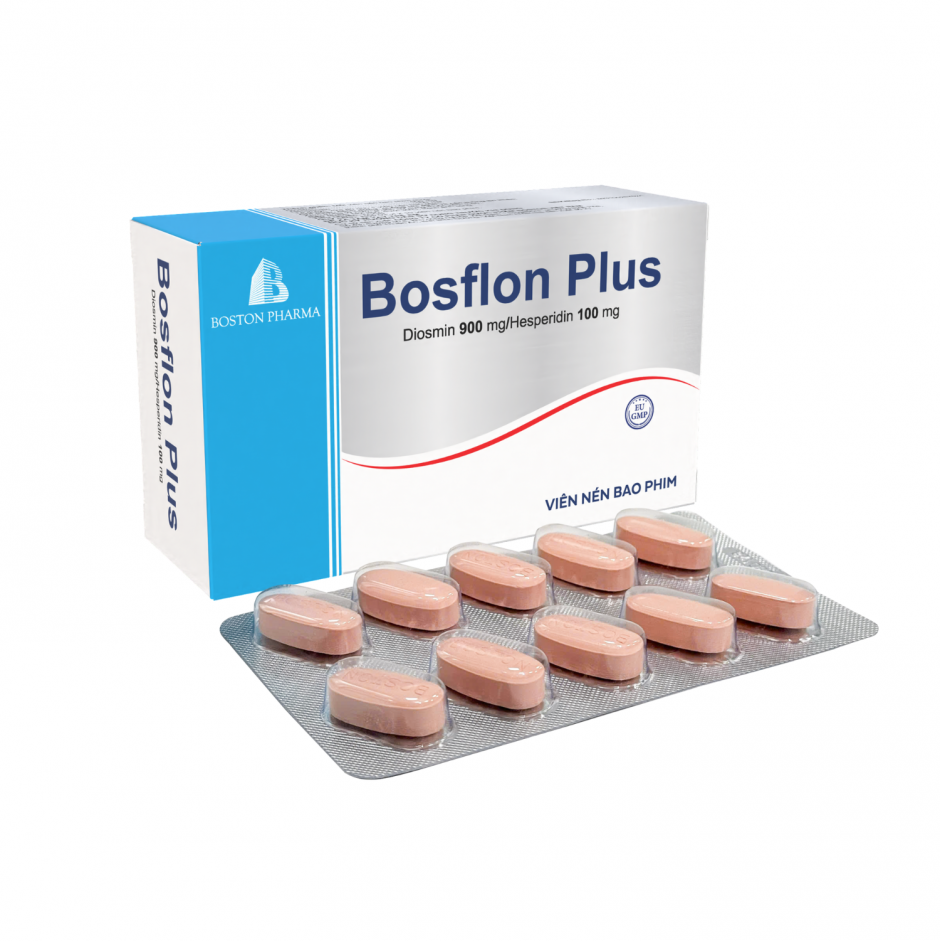
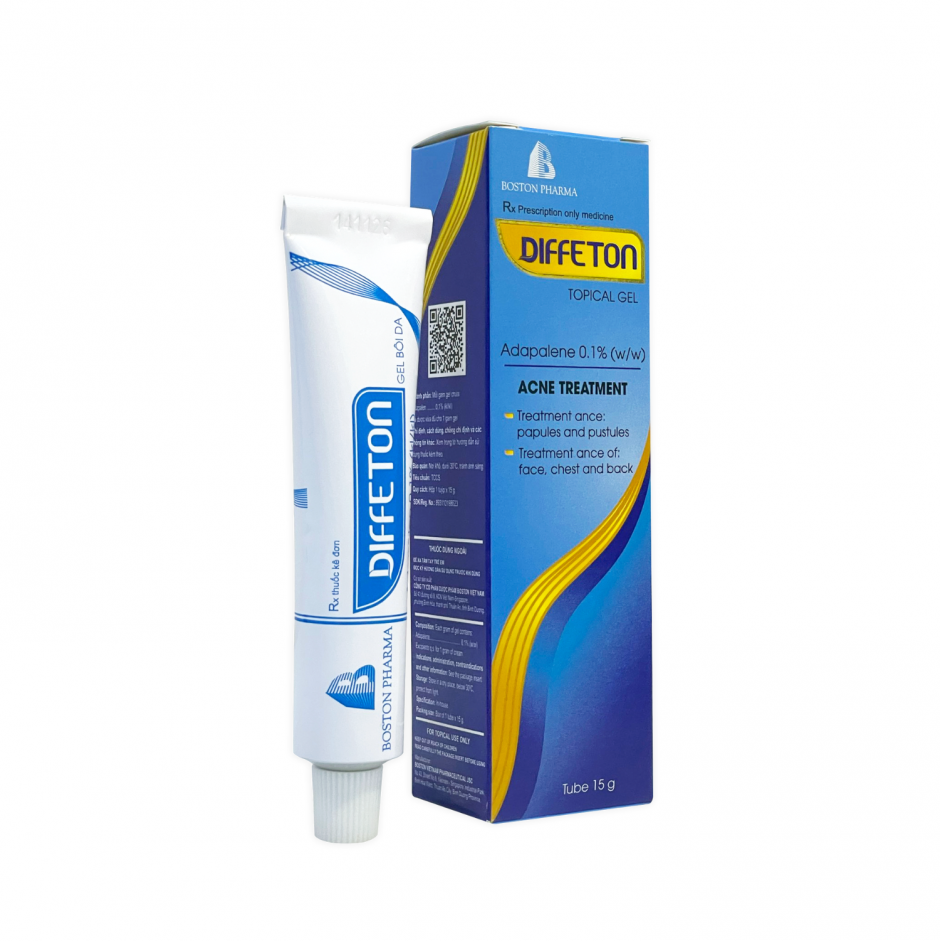
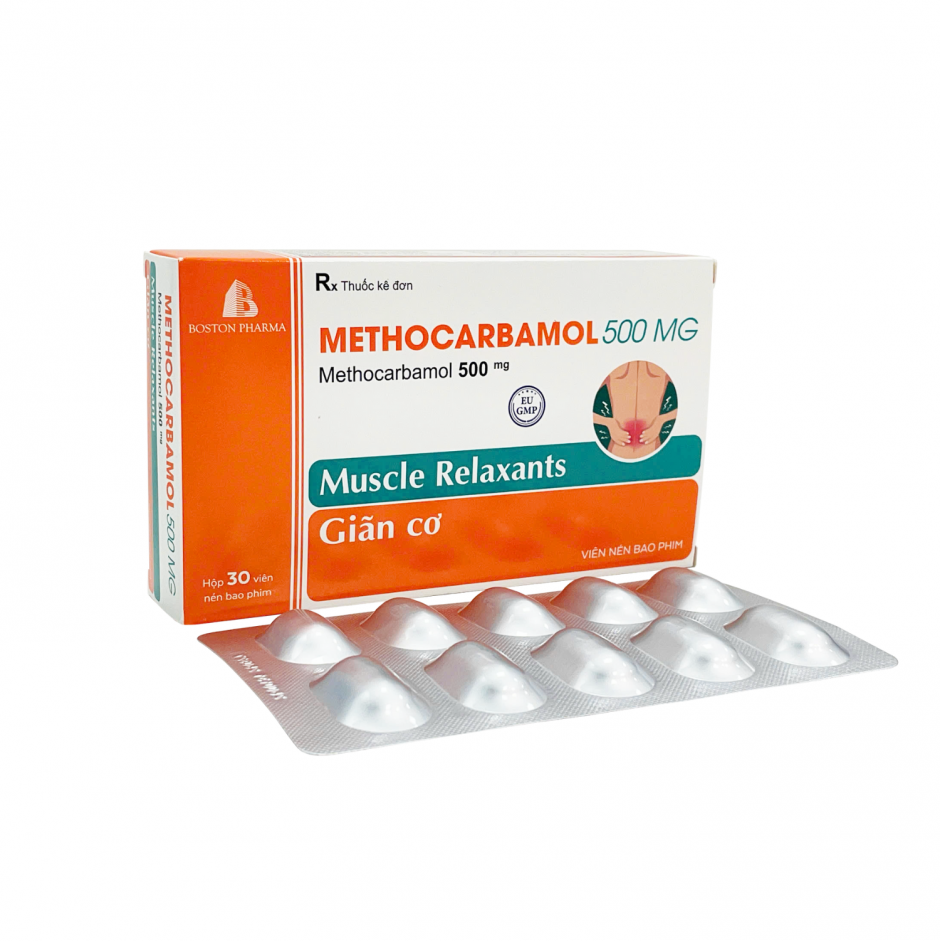
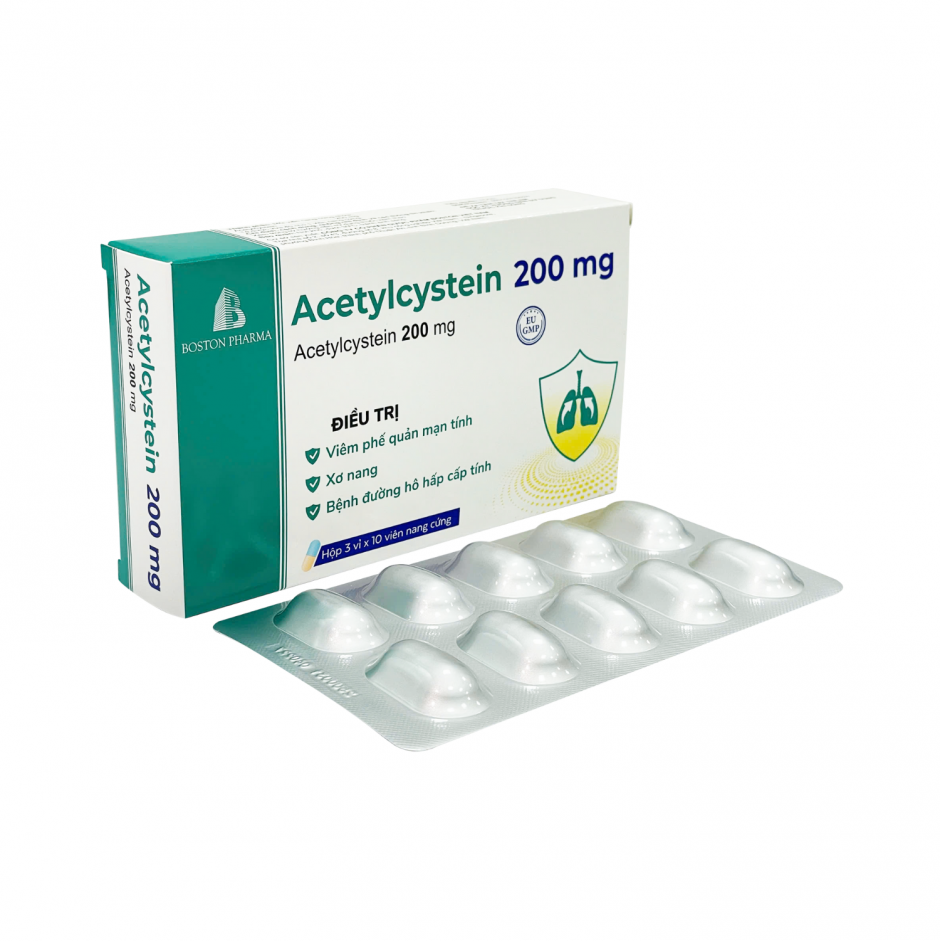
Factory: No. 43, 8th Street, Vietnam - Singapore Industrial Park, Binh Hoa Ward, Ho Chi Minh City, Vietnam. Phone: (0274) 3769 606
Office:
- At Lisbon Capital (Portugal): Campo Grande nº 28, 3.º C, - lisbon, district of Lisbon, municipality of Lisbon, parish of Alvalade.
Phone: 1900 1910
- At Ha Noi City: No.38 Duc Giang Street, Viet Hung Ward, Ha Noi City, Vietnam.
Phone: (024) 7305 0606
- At Da Nang City: No.01, Ham Trung 4 Street, Hai Van Ward, Da Nang City, Vietnam.
Phone: (023) 6730 3737
- At Nha Trang City: No.29, Thai Thong Street, Nam Nha Trang Ward, Khanh Hoa Province, Vietnam.
Phone: (025) 8730 3969
- At Ho Chi Minh City: 4th Floor, Flemington Tower, 182 Le Dai Hanh, Phu Tho Ward, Ho Chi Minh City, Vietnam.
Phone: (028) 3838 0606
- At Can Tho City: No.S2-3-4-5, 10th Street, Lot 11C RIVERINE Cantho City, Hung Phu Ward, Can Tho City, Vietnam.
Phone: (029) 2730 5959